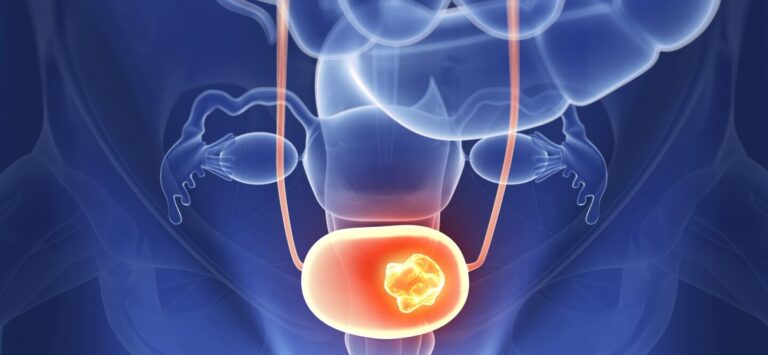
FDA recently approved the first gene therapy to treat high-risk bladder tumors that have not spread to muscle.
In a news release, the agency described nadofaragene Firadenovec-vncg as a nonreplicating vector-based genetherapy for adults with non-muscle-invasive urethral cancer (NMIBC), who do not respond to immunotherapy using bacillus Calmette-Guerin. According to the FDA, about 75% to 80% (or more) of newly diagnosed bladder tumors do not invade muscle. However, they are associated with a 30%-80% risk of recurrence, progression to metastatic and invasive cancer.
BCG, a type of bacteria is often used to treat NMIBC in adults to reduce the chance of recurrence. The FDA stated that patients with bladder cancer who do not respond to BCG have few options.
In a phase 3-trial, 157 patients were evaluated with high-grade NMIBC and BCG non-responsiveness. The treatment was administered every three months through a urinary tube for up to a year or until the toxic effects were too severe or NMBIC returned. Overall, 51% had a complete reaction, and no cancerous signs were seen during cystoscopy, biopsied tissues or urine. In the group of patients who responded positively to gene therapy, at least 46% showed a complete response lasting for more than a year. The results of the trial were published in January 2020.
The FDA reported that the most common side effects associated with this treatment included bladder discharge, fatigue and bladder spasm. Other adverse reactions include urinary urgency, bloody urine, fever, chills and chills.
Steven Boorjian MD, chair of Mayo Clinic’s urology and the lead investigator for the phase 3 trial on the new treatment, stated in a press release that patients with BCG-nonresponsive NMIBC had historically limited treatment options. Adstiladrin’s approval is a significant advancement in the current treatment landscape, and offers a new treatment option to patients.
Ferring Pharmaceuticals of Saint-Prex in Switzerland received FDA approval for a treatment that had previously been designated as a breakthrough therapy and a priority review. Ferring Pharmaceuticals announced that the new gene therapy would be available commercially in the US by the second half 2023.
According to the American Cancer Society, in the US, there will be 81 180 new bladder cancer cases and 17 100 deaths due to this disease by 2022. The American Cancer Society notes that bladder cancer is the fourth-most common cancer among men. However, it is rarer in women.
,