Rivermark Medical, a medical device company dedicated to the development of first-line devices for benign prostatic enlargement (BPH), has announced the successful use of its FloStent System in the RAPID-1 FIH study. The procedures were performed by Dr. Ingrid Perscky Arravanti, Principal Investigator (PI), and were supervised by Dr. Dean Elterman, Primary Clinical Advisor at Pacifica Salud Hospital in Panama City. The study evaluated the safety and effectiveness of the FloStent System.
“I’m happy to have been a part the first cases and am still very impressed with the performance of FloStent System. I was able position and deploy the implants using standard equipment and familiar endoscopic techniques,” said Dr. Ingrid Perscky Arravanti. She is a urologist, Clinical Professor Ad Honorem and President of the Global Men’s Health Foundation. This therapy is very attractive because it provides immediate relief from BPH symptoms, without the discomfort and inconvenience of conventional treatments.
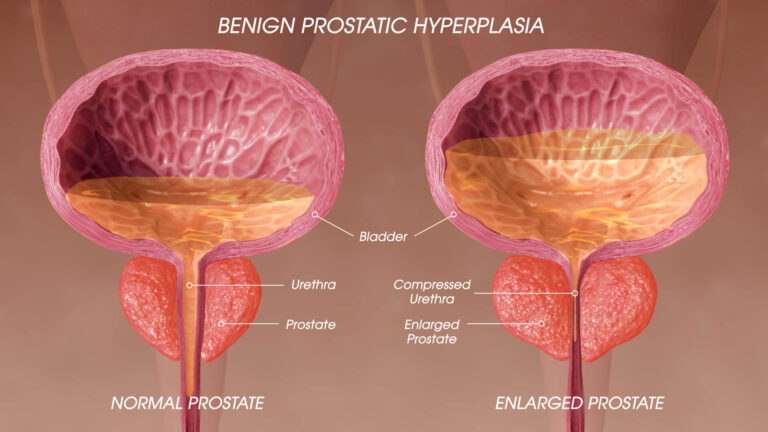
The FloStent System was designed to be a first-line treatment device for BPH. The outpatient procedure involves placing a nitinol catheter during a flexible cystoscopy. There is minimal recovery time for the patient and there is no need for an urethral cath. The stent gently opens the prostatic urethra to restore normal urination, improve sexual function and overall patient quality of life. The device can be easily removed or repositioned if desired.
Dr. Dean Elterman is a leading Urologist at the University of Toronto Division of Urology. He said, “I was impressed by the ease of the FloStent procedure and the immediate relief of BPH symptoms.” The ability to perform the FloStent procedure quickly with common flexible cystoscopes allows urologists to offer this device in a variety outpatient settings.
BPH, or benign prostatic hyperplasia (BPH), is caused by an enlarged prostrate. Over 200 million men worldwide are affected by this condition, which can cause uncomfortable symptoms like frequent urination and urinary tract infection (UTI). About 1 in 3 men older than 50 years old suffer from this condition, which impacts their quality of life. Current treatments have many drawbacks, including a lack of symptomatic relief, sexual dysfunction, infection risk, and discomfort during recovery.
Adam Kadlec M.D. is the CEO of Rivermark Medical, and a practicing Urologist in Milwaukee, Wisconsin. He said that today marks an important landmark as Rivermark transitions to a clinical-stage business. We are thrilled to be able offer BPH devices that are the easiest and most affordable in the world. This is a minimally invasive, first-line outpatient treatment, which can be done with existing equipment. It offers BPH patients reliable relief immediately. We look forward to building on the momentum we gained at FIH by gathering more clinical evidence demonstrating the benefits of the FloStent System in the months ahead.
,