Stanford Medicine researchers have made a discovery that could pave the road for a prophylactic nasal spray or “morning-after” spray to help prevent infection.
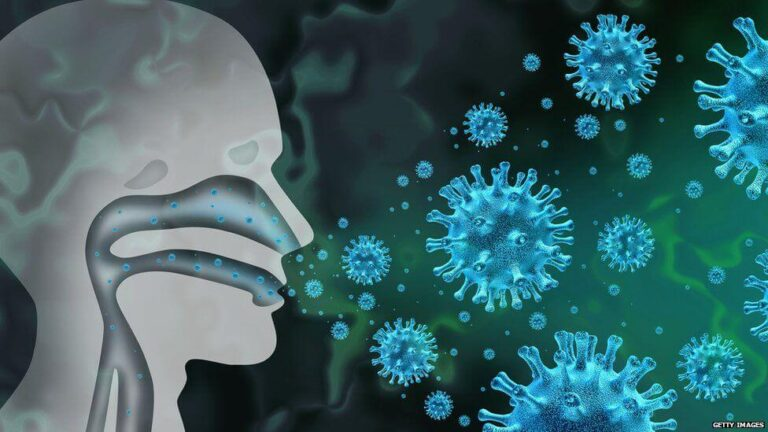
Someone just coughed in your face. On a flight. At a dinner party. In the supermarket queue
There should be a nasal spray for the morning after that can stop respiratory viruses from colonizing your nose and throat.
In a study that was published in Cell in print, Peter Jackson, Ph.D. a Stanford Medicine Professor of Pathology, Microbiology, and Immunology, and his co-workers brought this possibility closer to reality. They pinpointed the routes taken by SARS-CoV-2 (the COVID-19) to enter and leave cells in our nose cavity. Jackson stated that “our upper airways serve as a launchpad for not only infection of the lungs, but also for transmission to other people.”
Jackson and Raul Andino share senior authorship on the study, which is the first to describe COVID-19 nose infection in molecular details. Both are professors of microbiology, immunology, and UC San Francisco. Former Stanford Medicine postdoctoral fellow Chien-ting Wu (Ph.D.) and graduate student Ran Chang are the lead authors, as well as UCSF postdoctoral researchers Peter Lidsky (Ph.D.) and Yinghong Xiao (Ph.D.).
SARS-CoV-2 must attach itself to cilia on the cell surface in order to penetrate nasal epithelial tissues. Chien-Ting Wang
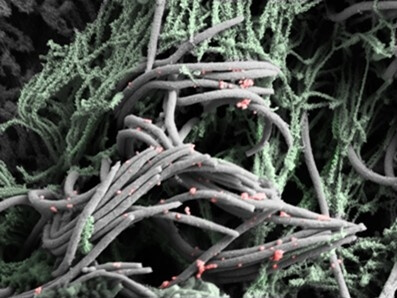
Nose and Airway
Nasal epithelium is lined by epithelial tissues, primarily consisting of basal cells and goblet-shaped cells. Multiciliated cells make up about 80%. Multiciliated cell forms a barrier that prevents viruses from entering the nasal airway.
Jackson and his co-workers focused on two structures that are found on multiciliated cells of the epithelium: microvilli and cilia. Both structures are well-known, but neither has been previously implicated in the way the virus enters or leaves the cells that line the airway.
Cilia are spaghetti like appendages that sprout from the surfaces facing outwards of different cells. One nasal epithelial cells may have up to 400 whip-like strands in its nasal cavity. They all beat together. The mucus is then covered with a thin coating of a mucin protein, which is closely related to mucus’ key protein.
Jackson explained that mucin molecules can form a mesh similar to a three-dimensional elastic chain-link fence. This mesh prevents larger viruses like SARS-CoV-2 getting into cells in the upper airways. The mucus coat traps viruses, bacteria, debris from the environment, and junk that breaks down cells, while keeping underlying tissues moist.
The upper-airway epithelial cells poke through the mucus, and their synchronized beats generate a wave, which pushes mucus along like a river.
Microvilli are another feature that is common to all animal cells. These tiny spikes extend from the surface of the cell like little fingers. Microvilli are able to transport vesicles and subcellular particles.
A virus infection that is still in its early stages
Jackson and his colleagues used a tissue culture technique to create what they called airway epithelial organismoids that mimic normal airways. These organoids, while lacking blood vessels or immune cells, otherwise accurately reproduce the architecture of nasal epithelium. This includes an intact mucus and mucin layer, and multiciliated, well-developed cells.
Scientists inoculated cultures by incubating the SARS-CoV-2 in the same dish. They used light and electron microscopes and immunochemical staining to monitor the epithelial cell for viral entry and replication.
Only cells with cilia became infected. The virus attaches to the cilia only at first, as shown by electron microscopy. Six hours after SARS-CoV-2 was incubated on organoids, the virus particles were dotted along the sides of the cilia from the tip down. Only a few cells were replicating the virus 24 hours after the inoculation. Massive replication took 48 hours.
SARS-CoV-2 infects a nasal epithelial cells (red), which induces changes to the stalk-like structures on their surface called microvilli. These grow into branching trees-like structures, which poke through extracellular mucus (not shown), and release viral progeny attached to them to spread to other cell types. Chien-Ting Wa
SARS-CoV-2 also needs at least a day to replicate in full force.
By knocking down the levels of a critical protein for cilia production in nasal epithelial cell, SARS-CoV-2 was severely slowed.
Jackson stated that “it’s clear the human ciliated nose epithelial cell is the primary entry point for SARS-CoV-2 into nasal epithelial tissues.”
Researchers treated airway organoids using a mucin selective enzyme to break down the mucin network mesh. They suspected that the delay was due to the mucus and mucin barrier that the virus must cross. The researchers found that it accelerated virus entry from “barely detectable” to “easily detectable” at 24 hours. Jackson concluded that the elimination of mucin in the mesh prevented the mesh blocking SARS-CoV-2 infections.
Mucus flow is affected by a rare condition called primary ciliary Dyskinesia. Patients with this disease have a compromised ciliary beat or ciliary that is not in sync.
The viral attachment to the cilia of airway organoids derived from these patients was similar to that observed in normal cells. Cell infection rates at 24 hours after inoculation were similar to normal infected cell levels. On the cell surface, normal-looking microvilli were bristled.
At 48 hours, SARS-CoV-2 infected far fewer total cells. It could only infect the cells immediately around it. This suggests that, once SARS-CoV-2 starts replicating inside infected cells the virus depends on mucus to spread through the upper airways.
Jackson co-authored a study in May 2020 Nature Communications that showed ACE2, the classic SARS CoV-2 binding cell-surface molecule or receptor, was concentrated on the cilias of nasal epithelial cellular. This receptor was found to be responsible for SARS-CoV-2 binding epithelial cells via the new cell study.
Jackson explained that the virus could then pass the mucus/mucin barrier by either skipping along the side of cilia in a hopscotch fashion, until it reaches the main body of the cell, where it will fuse with the membrane and climb in, or by squeezing its way through the cilium to ride an internal freight elevator to the cell body.
He said that once the virus has passed through this barrier, it can reproduce freely in cells below.
Researchers also discovered that SARS-CoV-2 once inside the cells, activates intracellular enzymes, causing microvilli, which are like crazy cactus, to grow and branch out, up until their tips protrude above the mucus membrane. They increase in number. Within 24 hours of the inoculation, altered microvilli that were normally less than half the size of cilia have grown into large, branching structures, the same size as cilia. They are decorated with viral particles which can be pushed into the mucus layer and spread to other cells.
Researchers identified enzymes that were massively activated by SARS-CoV-2 infections and responsible for the transformation of microvilli. The researchers inhibited these enzymes, which stopped the aberration and reduced the spread of virus to other cells.
All the ants can be bound with one spray?
Jackson and his collaborators had similar results incubating airway organoids either with the respiratory syncytial viruses, which are on the rise right now, or with the parainfluenza viruses that are less common. They also did the same with BA.1, the variant of the strain omicron.
Omicron, which is more infectious, infected multiciliated airway cells faster than the older strain that was used in the other SARS CoV-2 experiments. Even with this highly contagious variant, blocking viral entry or exit from airway cells was effective.
Jackson suggested that these viral entry mechanisms could be a common property of respiratory viruses.
These findings could lead to a new nasally administered drug that would prevent unknown respiratory viruses, such as those you might encounter in a pandemic, from settling into your nose and throat.
Jackson said that substances used in the experiments could be optimized to use, for example, in nasal sprays shortly after an exposure to a respiratory virus or as a prophylactic.
He said that by preventing viral entry, exit or spread, our immune systems would catch up, arrive in time and stop the full-blown illness.
The work was also supported by researchers from UCSF and Jikei University School of Medicine, Tokyo, as well as the Texas Biomedical Research Institute.
The study was supported by the National Institutes of Health, Stanford Diabetes Research Center (fast grant), Defense Advanced Research Project Agency and Stanford Respond Innovate Scale Empower.
,