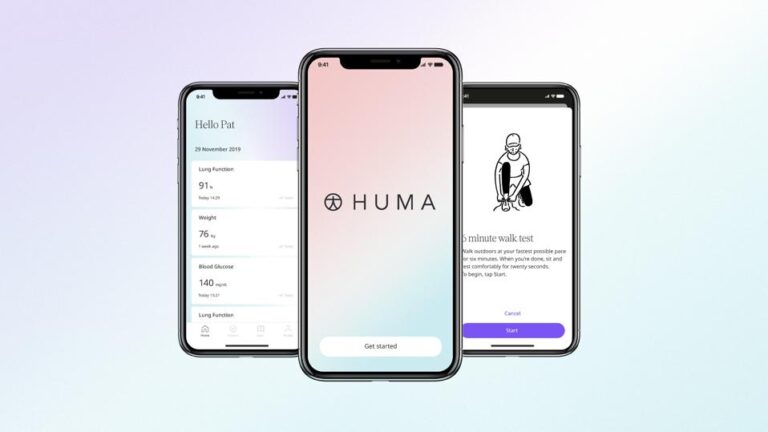
- Over 90% of the 2,524 participants who were invited to take part in a virtual COVID-19 research study run by Cambridge researchers downloaded and registered the study app
- At least 75% of participants performed digital biomarker assessments, such as temperature, oxygen saturation, and resting heart rate at least once a week.
- Participants aged 45-70 years took part in the program for an average 8 months.
Huma Therapeutics Limited (Huma), a leading digital health company in the world, has announced the results of an innovative study conducted in collaboration with Medical Research Council Epidemiology Unit, University of Cambridge. The study showed that participants who used Huma’s platform for clinical trials had sustained high levels of engagement during a fully remote observational COVID-19 study.
The study published in the Journal of Medical Internet Research involved 62.61% of participants (2524/4031) from the longitudinal Fenland Study. This is the largest population-based research study that has been conducted to date.
Ninety-two percent (2277/2544) of the participants completed the app-based registration process and signed a consent electronically. The study app was available for iPhone and Android, and each participant received a digital thermometer and pulse oximeter to measure blood oxygen levels.
The biomarkers below were recorded by the participants after receiving remote assistance.
- Blood oxygen saturation (three time per week)
- Body temperature (three times a week)
- resting heart rate (three times per week)
- Activity levels (measured passively).
- Answer monthly questionnaires
- Tests for coronavirus antibodies using blood spot samples
The study app was easy to use, and participants were able to quickly report symptoms and measurements. The participants took part in this observational study at least for six months, and they completed all measures until told to stop. There was only a minimal drop in engagement during the study period. In average, participants used the app for 34,5 weeks (7.9 month) and only 2.5% withdrew from the study. The engagement rate was higher among participants over 65.
Dr. Arrash Yeassaee is the Global Clinical Director of Huma. He said, “Huma’s commitment to building robust clinical evidence and scientific proof for its technology is evident.” This study has shown a high level of engagement, which is encouraging. Huma’s user-friendly clinical trials technologies have the potential to transform health research in populations by increasing accessibility and reducing burden on participants. This type of data collection can be extremely valuable in understanding health and disease processes and gathering insights to make a real difference to people’s life.
Dr. Kirsten Renni, Senior Research Associate, MRC Epidemiology Unit, and expert in quantitative measures for physical activity and nutrition, led the study. She said, “Enrolment in cohort-based observational research is a challenge, and participation in these studies has declined in recent years.” We saw that not only was there a high rate of enrolment, but also a high level of engagement. This helped us to create a checklist that other researchers can use.
The COVID-19 epidemic has sparked interest in digital health solutions that allow remote monitoring of health. These technologies are beneficial for patients with chronic and acute conditions. However, their use in population-based research is less well known. It is difficult to recruit and retain participants over many months or years.