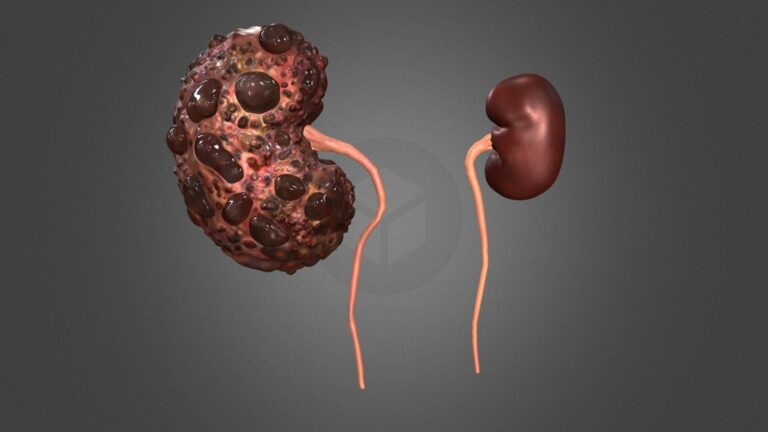
The National Institutes of Health supported a research team that developed a new method to better understand polycystic renal disease (PKD), affecting millions of people worldwide. Scientists used two methods to model the disorder – organs-in-a dish and organs-on-a chip technologies – to demonstrate the role glucose, a common sugar found in blood, plays in forming PKD cysts. The findings, published in Nature Communications could help develop better methods to test and treat PKD and other diseases.
Organoid or an organ-in-a dish is a miniature of a lab-grown organ. It can mimic the key features of an organ’s function and structure. Organs-on a chip, or tissue-chips, are 3-D models that contain channels and live cells to simulate organ and tissue structures and environments. The National Center for Advancing Translational Sciences’ (NCATS) programs are developing both technologies and human cell-based methods to study diseases and better predict whether drugs would be safe or toxic for humans.
In PKD the tiny tubes in the kidneys (tubules), expand like water balloons over many years, forming fluid sacs. These cysts or sacs eventually crowd out healthy tissues, causing kidney problems and even kidney failure. Many of the genes responsible for PKD have been identified, but many aspects of the disease remain unknown, such as how cysts form.
The University of Washington School of Medicine’s Benjamin Freedman Ph.D. led the research. The results of animal models can be helpful, but translating them to humans has been difficult.
Freedman, Jonathan Himmelfarb M.D. and their Seattle colleagues decided to combine organoid tech with a platform of tissue chips. Scientists believed that fluid flow was important for the development of cysts but had no way to test the theory using organoids.
Freedman explained that “in kidneys, fluid always flows through the tubules. At any given time the kidneys are carrying about 25% of body fluid.” We can’t replicate this system in a dish because fluid must move through kidney structures. The next step was to use microfluidics in tissue chips.
The mini-kidney tubes have sugar receptors in the upper left (red) and are arranged to form polycystic renal disease cysts that face outwards (center). These cysts swell when they take in sugar (green lower right). University of Washington
Freedman’s group demonstrated that exposing a PKD organoid on a chip model to a mixture of water, sugars, amino acids, and other nutrients led to cysts expanding relatively quickly. The cysts absorbed glucose and drew water from fluids passing over them. This caused the cysts to grow. Although glucose is absorbed by kidneys in general, it has not been linked to cyst formation.
It wasn’t surprising that the cysts were able to absorb glucose. However, it was surprising that their dependence on it. Freedman explained that the new theory is a different way to think about how cysts develop.
Scientists found that mice cysts took up glucose when they added fluorescent glucose. “We believe the tubules in the mouse are absorbing fluid, just as in organoids.” Freedman explained that as the kidney grows, the cysts develop.
Understanding the mechanisms behind PKD may lead to new treatment options. The research team found that compounds that block glucose transportation prevented cyst growth. Freedman said that glucose inhibitors were being developed for kidney diseases other than cystic kidney disease.
The NCATS Office of Special Initiatives director, Danilo Tagle Ph.D. said that the researchers had shown how simulating fluid movement is crucial to make this system more similar to the environment of the kidney in PKD. Combining the two technologies will make tissue chip technology more flexible for drug discovery and development, and allow researchers to benefit from the strengths of both platforms. It is very promising to study other diseases in new and innovative ways in the near future.
The research was funded by NCATS, the National Institute of Diabetes and Digestive and Kidney Diseases, and the National Heart, Lung, and Blood Institute, through NIH Grants UG3TR002158, UG3TR003288, UG3TR000504, R01DK117914, UC2DK126006, and UG1HL152401.
,