- Rybelsus is the first and only GLP-1 analog available in oral form. It can be used as an initial treatment for people with type 2 diabetes, together with diet and physical activity.
- Rybelsus, since its approval in 2007, has been prescribed by hundreds of thousands to improve glycemic management along with diet and physical activity.
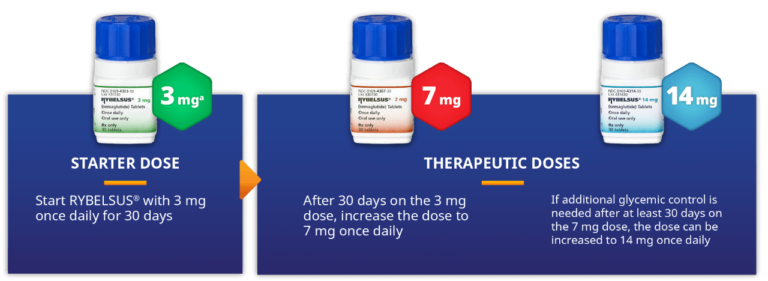
The U.S. Food and Drug Administration has approved an update to the label for Rybelsus tablets (7 mg or 14mg) that allows adults with type-2 diabetes who have never taken diabetes medication before, to use it as their first line treatment. The FDA has approved a label update that removes an old restriction of use stating the medication shouldn’t be used to treat patients with type-2 diabetes as the first treatment. Rybelsus was approved by FDA in 2019 and is the only GLP-1 analogue in pill form. It is used in conjunction with diet and physical exercise to improve the glycemic management of adults with type 2 diabetics.
Dr. Aaron King is a Family Medicine and Diabetic Specialist. He said that the removal of the restriction on use was an important step for people with type 2 diabetics and allows Rybelsus be taken earlier. By taking Rybelsus before other diabetes medications, patients with type 2 diabetes can now begin their treatment earlier.
Rybelsus lowers blood sugar differently than other diabetes medications. It does so in three different ways: it increases the release insulin from the pancreas, decreases the release sugar from the liver and slows the process by which food leaves the stomach after consuming. Rybelsus comprises a unique co-formulation of semaglutide and an absorption enhancer called SNAC (sodium N-(8-[2-hydroxybenzoyl] amino) caprylate), which facilitates absorption of semaglutide in the stomach, making it possible to provide semaglutide as a pill.
Doug Langa is executive vice president and president of Novo Nordisk’s North America operations. He said that hundreds of thousands in the U.S. have received this medication as part of a type 2 diabetic treatment plan to lower their A1C. Rybelsus is a key part of Novo Nordisk’s portfolio as it celebrates 100 years of innovation and commitment in diabetes care. Rybelsus has made history by being the first GLP-1 receptor oral agonist, and helps fuel our mission to help improve the lives of people with diabetes.
Novo Nordisk collaborates with health insurance companies to ensure Rybelsus is covered by all insurance plans and that patients have access to it. Commercially insured patients who are eligible may pay as low as $10 for an order of Rybelsus that lasts from one to three months.
Important Safety Information
What is RYBELSUS?
RYBELSUS tablets (7 mg or 14mg) are a prescription medication used in conjunction with diet and physical activity to help improve blood sugar levels (glucose).
- It is unknown if RYBELSUS may be used by people with pancreatitis
- RYBELSUS should not be used by people with Type 1 diabetes
- It is unknown if RYBELSUS can be used safely and effectively by children younger than 18 years.
What are the most important facts I need to know about RYBELSUS
RYBELSUS can cause serious side effects including:
- Thyroid cancer, as well as possible thyroid tumors. Inform your doctor if you experience a lump in your neck or throat, difficulty swallowing or shortness breath. These symptoms may indicate thyroid cancer. RYBELSUS, and similar medicines, caused thyroid tumors in rodents. It is unknown if RYBELSUS can cause thyroid tumors in humans or a form of thyroid cancer known as medullary Thyroid Carcinoma (MTC).
Use RYBELSUS only if you:
- If you or anyone in your family has ever suffered from MTC or if you suffer from an endocrine condition known as Multiple Endocrine Neoplasia Syndrome type 2 (MEN2)
- You have had a severe allergic reaction to semaglutide, or any other ingredient in RYBELSUS.
Tell your healthcare provider if any of the following conditions exist before using RYBELSUS:
- You may have problems with your kidneys or pancreas
- Have you ever had vision problems associated with diabetes?
- Are pregnant or intend to become pregnant. It is unknown if RYBELSUS can harm your unborn child. Stop using RYBELSUS 2 months before your planned pregnancy. Speak to your healthcare provider to find out the best ways to manage your blood sugar levels if you intend to become pregnant, or if you are already pregnant.
- Are breastfeeding or plan on breast-feeding. Breastfeeding during treatment with RYBELSUS is not recommended.
Inform your healthcare provider of all medicines that you are taking. This includes prescription and over-the counter medicines, vitamins and herbal supplements. RYBELSUS can affect how some medicines work, and some medicines can affect the way RYBELSUS functions.
How should I take RYBELSUS?
- Follow the instructions of your doctor when taking RYBELSUS.
- Take RYBELSUS orally on an empty stomach as soon as you wake up. Drink a small amount of water (nomore than 4 ounces).
- Avoid crushing, chewing, or splitting RYBELSUS. Do not split, crush or chew.
- After 30 minutes you can drink, eat or take oral medications.
- If you forget to take a dose, go back to the regular schedule.
What are possible side effects from RYBELSUS?
RYBELSUS can cause serious side effects including:
- Pancreatitis, or inflammation of the pancreas. If you experience severe abdominal pain that won’t go away with or without nausea, stop using RYBELSUS immediately and contact your healthcare provider. The pain may radiate from your stomach to your back.
- Changes to vision. Inform your healthcare provider if there are any changes in your vision while taking RYBELSUS.
- Hypoglycemia. You may have a higher risk of low blood glucose if you take RYBELSUS along with other medicines that cause low blood glucose, such as sulfonylureas or insulin. Low blood sugar can cause dizziness, lightheadedness, blurred or cloudy vision, anxiety, irritability, or mood changes. Other symptoms include sweating, slurred or garbled speech, hunger or confusion, weakness or drowsiness.
- Kidney Failure. Dehydration can worsen kidney problems in people with kidney problems. Drinking fluids can help you avoid dehydration.
- Severe allergic reactions. If you experience any of the following symptoms, stop using RYBELSUS immediately and seek medical attention. These include swelling of your lips, face, tongue or throat, difficulty breathing or swallowing, severe rash or itching, fainting, feeling dizzy, or a rapid heartbeat.
- Gallbladder Problems. Gallbladder issues have occurred in some people taking RYBELSUS. You should contact your doctor immediately if you experience any symptoms of gallbladder issues. These include pain in the upper abdomen (abdomen), jaundice (yellowing of the skin or eyes), fever and clay colored stools.
RYBELSUS can cause nausea, stomach pain (abdominal), diarrhea, reduced appetite, constipation, and vomiting. RYBELSUS can cause nausea, vomiting and diarrhea.
You can find useful links: Prescription information and Medication Guides for RYBELSUS (r)
Visit Rybelsus.com for more information. Visit RybelsusPro.com for health care professionals.